Prof. Dr. Borna Relja

Prof. Dr. Borna Relja
Former Project Leader (04/2020 - 02/2023)
Project 10
P10-1: Maladaptation of the hepatic barrier in alcohol-induced liver injury
Aleksander NowakPhD Student
Borna ReljaProject Leader |
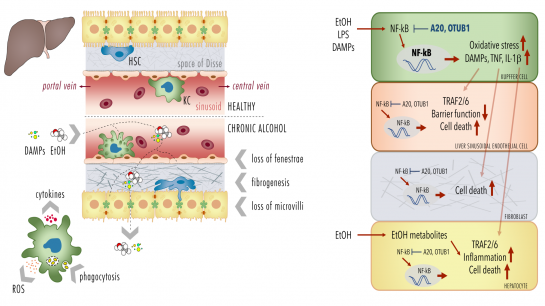
Alcoholic liver disease (ALD) as one of the predominant causes of liver-related morbidity and mortality worldwide encompasses a spectrum of liver injury ranging from simple steatosis to steatohepatitis, fibrosis, and finally cirrhosis. The pathogenesis of this multifactorial disease in-volves both hepatic non-parenchymal and parenchymal cells (hepatocytes). The project focus on functional studies in a murine model and isolated primary Kupffer cells (KCs), liver sinusoidal endothelial cells (LSECs) and hepatocytes from mice being chronically fed with a Lieber-DeCarli diet containing alcohol (ethanol, EtOH) or an isocaloric control diet. Following induction of the early stage of ALD, comparative analyses will be conducted in the murine model, scrutinizing hepatic barrier integrity and systemic and local inflammation. Herein chemokines, cytokines, DAMPs, leukocyte activation and hepatic infiltration via immuno-histology, flow cytometry, organ histopathology will be analysed (cooperation with Project 7, Project 8 and Project 9). Further, loss of fenestrae, fibrogenesis, necroptosis, apoptosis, pyroptosis, and oxidative burst as well as phagocytosis by KCs in different cell types will be investigated. In addition, the NF-κB activity and cellular responses (cytokine release, cell survival) of each isolated primary cell type (KCs, LSECs and hepato-cytes) will be studied (cooperation with Project 1). Cells will be isolated by enzymatic digestion of liver tissue and gradient centrifugation. For the isolation of cells selective adherence behaviour (KCs), and subsequent F4/80 (KCs), CD45 and CD31 (LSECs) or ASGPR (hepatocytes) will be used as signature expression markers. NF-κB signaling is regulated by a variety of posttranscriptional modifications (PTMs), including covalent conjugated ubiquitin. Deubiquitinylating enzymes (DUB) cleave ubiquitin from substrate proteins and are hence key regulators of the NF-κB sys-tem. DUBs A20 or OTUB1 regulate/terminate TNF- or IL-1β-induced NF-κB activation, respec-tively, suppressing inflammation and oxidative stress, but also DNA repair and cell death. To determine the causality of DUBs, selected DUBs will be knocked down (A20 and OTUB1) and the consequences of chronic exposure to EtOH, or stimulation with endotoxin or DAMPs on NF-κB activity, cytokine release, inflammasome activation and cell survival (immunoblots, ELISA, FACS) will be evaluated in isolated primary cells (KCs, LSECs and hepatocytes) and different hepatic human cell lines (human Kupffer cells, HLSEC/ciJ LSECs, HepG2 and AML12 hepato-cytes etc. in cooperation with Project 1 and Project 7).
Proposed mechanisms of A20 and OTUB1-mediated maladaptation of the hepatic barrier loss. |
Photos: by UMMD, Melitta Schubert/Sarah Kossmann
MD3: Role of A20 in lung barrier breakdown after trauma
|
|
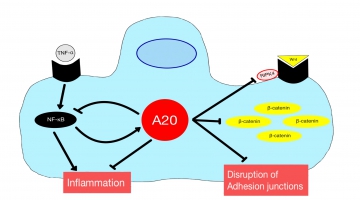
Traumatic injuries as femur or pelvic fracture followed by hemorrhagic shock often lead to Remote Lung Injury (RLI), which forms the basis for the development of secondary complications such as Acute Respiratory Distress Syndrome (ARDS), contributing to the major causes of global mortality. However, the exact mechanisms of the RLI development still remain obscure. This project focuses on the disclosure of the pathophysiology, with special emphasis on the NF-kB and the Wnt signaling pathways and the impact of deubiquitinase A20 on both pathways. We hypothesize that the deubiquitinase A20 is downregulated after trauma, leading to an upregulation of Wnt signaling and the accumulation of ß-catenin by repealed inhibition of RIPK4. This leads to the disruption of adhesion junctions and ultimately to the breakdown of the lung cell barrier. Additionally, we claim that the effect of A20 on the NF-kB signaling pathway is downregulated as a negative feedback loop, leading to an increased inflammation within the lung tissue. Moreover, prior acute alcohol intoxication should have an immunomodulatory effect on NF-kB and the intratracheal administration of A20 should invert the effects described above. Therefore, both interventions could lead to a stabilization of the lung barrier, which would ultimately contribute to an improved outcome. In an in vivo model, mice receive a femur fracture in a mouse model followed by hemorrhagic shock. Additionally, ethanol is applied intragastrally and/or the A20 protein intratracheally prior trauma. Subsequently, the extent of lung damage is evaluated by measuring the total protein content in a bronchoalveolar lavage and the Lung Injury Score will be validated by lung histopathology. By using immunohistochemical stainings and quantitative protein analysis (ELISA, Western blot and flow cytometry) the extent of neutrophil and polymorphonuclear leukocyte invasion, local inflammatory changes (IL-6, CXCL1, CXCL5, TNF-a, NF-kB), lung damage (A20, RIPK4 and ß-catenin), apoptosis (M30 and active caspase-3) and pyroptosis (NLRP3) will be determined.
|
Photos: by UMMD, Melitta Schubert/Sarah Kossmann