AP2: Role of STAMBPL1 in cell survival in gastric mucosa
|
|
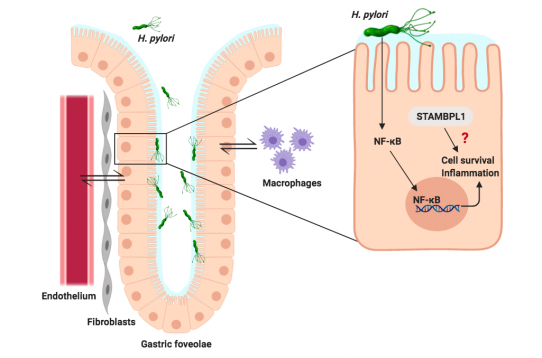
The human pathogen Helicobacter pylori, a risk factor for gastric diseases, induces the transcription factor NF-κB leading to a pro-inflammatory response and cell survival. These processes are regulated by a variety of deubiquitinylating enzymes (DUBs), which are overexpressed or mutated in a variety of cancer types. STAM-binding protein-like 1 (STAMBPL1, also called AMSH-2 and AMSH-LP) belongs to family proteins of JAMM DUBs that cleaves Lys63 ubiquitin linkage. However, the functions of STAMBPL1 and target proteins have not yet been understood. In order to scrutinize the roles of STAMBPL1, we aim to identify the mechanisms of STAMBPL1 function in H. pylori-infected gastric epithelial cells and at epithelial gastric barriers. In our functional studies, STAMBPL1 depletion causes apoptotic cell death accompanied by caspase 8 and PARP-1 cleavage as well as a decrease in the abundance of XIAP and surviving in non-stimulated HeLa cells, but not in non-stimulated AGS and NCI-N87 cells. Interestingly, H. pylori infection led within 16 h to the degradation of STAMBPL1 through K48 ubiquitinylation. Therefore, it is of interest to identify which E3 ligases regulate this process. Further, the molecular crosstalk of the infected epithelial barrier with the mucosa cells (fibroblasts and lymphangiogenic endothelial cells) and the consequences for the micromilieu will be studied. To investigate the roles of STAMBPL1 homeodynamics and dysregulation in gastric barrier function, we plan to analyze the influence of STAMBPL1 on the secretome of the micromilieu (fibroblasts and endothelial cells) in the gastric mucosa during H. pylori infection. Secreted factors which induce proliferation and survival will be identified and addressed in detail in co-culture system. The impact on angiogenesis of endothelial cells in response to the secretome of H. pylori-infected epithelial cells will be also studied. In order to study the maladaptive inflammatory processes, we plan to study the inflammation in STAMBPL1-depleted epithelial cells and the impact on the micromilieu using primary gastric organoid cultures (cooperation with MPI Berlin, University of Dresden). In long-term culture, the impact of immune response mediators on cell survival will be studied by qPCR, multiplex cytokine assay and monitoring of apoptotic cell death factors.
STAMBPL1 in cell survival and crosstalk with gastric mucosa during H. pylori infection. Roles of STAMBPL1 on cell survival, gastric barrier function and maladaptive response will be addressed in detail. |
Photos: by UMMD, Melitta Schubert/Sarah Kossmann